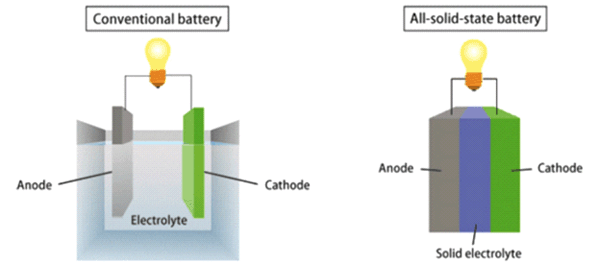
For application’s purpose, the battery technology which is common and is currently used is Nickel Metal Hydride (Ni-MH). The battery’s chemistry determines the capacity of the cell and the cell as how much electrical energy can be given by the battery. When an external source is connected with these electrodes, as a result of chemical reaction, electrons are released and therefore for tapping the current. The transfer of the ion between the electrodes is allowed by the dissociated salt. A negative (anode) and positive (cathode) electrode are in each cell, separated by an electrolytic solution, known as an electrolyte. Essentially, many electrochemical cells joined in parallel or series are known as a battery and they provide capacity and voltage. IntroductionĬathode is responsible for the positive charge and the anode is responsible for the negative charge and their division is carried out by an electrolytic solution most commonly known as an electrolyte. In various technologies, for instance, Hybrid Electric Vehicles (HEV) uses both, lithium-ion batteries and electric motor engines for increasing fuel efficiency. These are essential because all the electrode balancing and chemical charge balancing happens on the basis of these. Lithium-ion batteries are discussed here and about how and what sort of mechanics are involved while preparing the mechanics of cathode and anode materials. Cathode and Anode materials are a part of every battery because this is the main source of how the working of a battery is enhanced or properly stimulated.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
